| Theonellapeptolide-Id
Date: Dec.1999 Mtx-Val-D-mLeu-(-Thr-bAla-D-Leu-mIle-bAla-D- alloIle-mVal-mAla-bAla-D-Leu-malloIle-), 13-water hydrates Mtx=methoxyacetate, m=N-methyl, bAla=beta-alanine |
|
Chemical scheme is here.
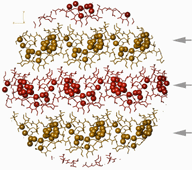
Left: Amphiphatic layer structure in the crystal. Colored balls represent the solvated water molecules and the arrows show the water-layers. Drawn radius is 35 A. |
Paper:
-
Tile: Amphipathic Structure of Hydrated Theonellapeptolide-Id, a Hydrophobic
Tridecapeptide Lactone from the Okinawa Marine Sponge
Theonella swinhoei
Authors: Mitsunobu Doi, Toshimasa Ishida, Motomasa Kobayashi, Yoshio Katsuya, Yushihiro Mezaki, Masahiro Sasaki, Akira Terashima, Taizo Taniguchi and Chikako Tanaka
Journal: Biopolymers 54, 27-34 (2000)
References of theonellapeptolide:
- Kobayashi, M., Lee, N. K., Shibuya, H., Momose, T. & Kitagawa, I. (1991) Chem. Pharm. Bull. 39, 1177-1184.
- Kobayashi, M., Kanzaki, K., Katayama, S., Ohashi, K., Okada, H., Ikegami, S. & Kitagawa, I. (1994) Chem. Pharm. Bull. 42, 1410-1415.
- Kobayashi, M., Kawazoe, K., Okamoto, T., Sasaki, T. & Kitagawa, I. (1994) Chem. Pharm. Bull. 42, 19-26.
- Kobayashi, J., Itagaki, F., Shigemori, H., Ishibashi, M., Takahashi, K., Ogura, M., Nagasawa, S., Nakamura, T., Hirota, H., Ohta, T. & Nozoe, S. (1991) J. Am. Chem. Soc. 113, 7812-7813.
- Kobayashi, M., Kawazoe, K., Katori, T. & Kitagawa, I. (1992) Chem. Pharm. Bull. 40, 1773-1778.
- Kitagawa, I., Lee, N.K., Kobayashi, M. & Shibuya, H. (1987) Chem. Pharm. Bull. 35, 2129-2132.
- Kitagawa, I., Lee, N.K., Kobayashi, M. & Shibuya, H. (1987) Tetrahedron,
47, 2169-2171.
Structure... - Doi, M., Ishida, T., Kobayashi, M., Deschamps, J. R. & Flippen-Anderson, J. L. (1999) Acta Crystallogr., Sect. C, 55, 796-798. (see orthorhombic [monomeric] structure)
- Bernardinelli, G., Jefford, C.W., Sakai, R. & Higa, T. (1992) Abstracts of Papers, 7th International Symposium on Marine Natural Products, p29-30, Capri, Italy.
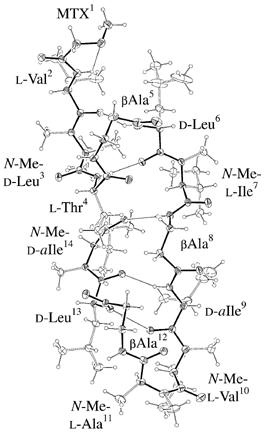
ORTEP-III (Burnett, 1996) drawing of Molecule 1. |
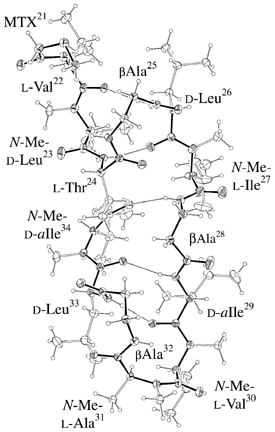
ORTEP-III (Burnett, 1996) drawing of Molecule 2. |
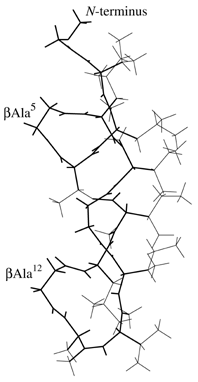 Side view of Molecule 1. Main chain is thick line and fairely winded. |
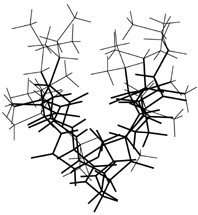 Side view of Molecule 2. Shape is like as a "bottom of ship". |
X-Ray Data Summary
| formula | 2(C70H125N13O16), 12.9H2O | ||
| weight | 3043.4 | ||
| symmetry | monoclinic | ||
| space group | P21 | ||
| Cell | Crystal | ||
| a | 11.9955(3) Ang. | description | block |
| b | 36.9917(10) Ang. | colour | Colorless |
| c | 19.7887(3) Ang. | size (mm) | 0.3x0.2x0.2 |
| alpha | 90.0 deg. | Dx (g/ml) | 1.069 |
| beta | 96.1623(15) deg. | F(000) | 3056 |
| gamma | 90.0 deg. | mu(CuKa) | 0.076 |
| volume | 8730.2(3) Ang^3 | wave length | 0.834 A |
| Z | 4 | temperature | 100(2) |
| Diffrn measurement | Refinementspace | ||
| device type | Rigaku RAXIS-4 | Flack | 0.5(11) |
| radiation source | SPring-8/BL24XU-A* | parameters | 2002 |
| index limit | 0~h~15, 0~k~46, -24~L~24 | restraints | 396 |
| theta (deg.) | 2.3-31.5 | R_factor_gt | 0.0865 |
| total reflections | 16300 | wR_factor_gt | 0.2492 |
| reflections(obs) | 14826 .gt. 2sigma(I) | dela rho_max | 0.802 e A^3 |
| Structure | delta rho_min | -0.428 e A^3 | |
| solution | SnB | shift/su_max | 7.069 |
| refinement | SHELXL-97 | Goodness of fit | 1.111 |
*The machine time of the synchrotron was given from Hyogo prefecture and Japan Synchrotron Radiation Research Institute with the proposal No. C99A24XU-005N.
Get PDB
Back to the structure index
