| Peptoid-Boc-Phe N-tert-Butyloxycarbonyl-N-benzylglycine 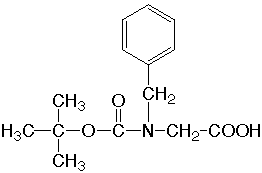 |
 This structer was produced by RasMol, and stucked by GifBuilder (Yves Piguet, 1997) |
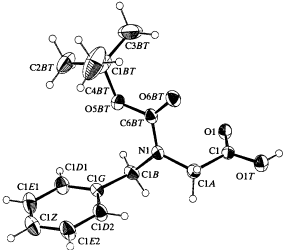 ORTEP-III (Burnett, 1996) drawing |
Peptoids, amino acid mimics, have the basic unit of N-substituted glycine derivatives and were designed as a new series of potentially bioactive compounds (Simon et al., 1992; Zuckermann, Kerr, Kent, & Moos, 1992). The side chain or functional group is bonded to the alpha-nitrogen, contrary to the usual side chain, bound to the alpha-carbon. The chemical structure is similar to that of a \b-amino acid, and metabolic stability, reduction of conformational constraint by chirality and wide variability of functional groups are expected (Figliozzi, Goldsmith, Ng, Banville, & Zuckermann, 1996). Relative to polypeptides, polypeptoids have their side chains shifted by one position along the backbone. A monomer derivative, N-tert-butyloxycarbonyl-N-benzylglycine (Boc-Nphe), was crystallized from ethyl acetate solution in the centrosymmetric space group P21/a. Paper: Mitsunobu Doi, Kazue Kinoshita, Akiko Asano, Ryuji Yoneda, Takushi Kurihara & Toshimasa Ishida Acta Crystallogr. Sect. C, 54, 1164-1165 (1998). |
X-Ray Data Summary
| formula | C14 H19 N O4 | ||
| weight | 265.30 | ||
| symmetry | monoclinic | ||
| space group | P21/a | ||
| Cell | Crystal | ||
| a | 13.323(2) Ang. | description | block |
| b | 7.8005(9) Ang. | colour | Colorless |
| c | 15.324(2) Ang. | size (mm) | 0.6x0.4x0.1 |
| alpha | 90.0 deg. | Dx (g/ml) | 1.181 |
| beta | 110.499(8) deg. | F(000) | 568 |
| gamma | 90.0 deg. | mu(CuKa) | 0.713 |
| volume | 1491.7(3) Ang^3 | ||
| Z | 4 | ||
| Refinement | Diffrn measurement | ||
| Flack | device type | Rigaku AFC5R/RU-200 | |
| parameters | 176 | decay | 0.7 % |
| restraints | 0 | index limit | 0-h-15,-9-k-0,-17-L-16 |
| R_factor_all | 0.0954 | theta (deg.) | 3.08 - 63.18 |
| R_factor_gt | 0.0679 | total reflections | 2533 |
| wR_factor_ref | 0.2884 | reflections(obs) | 1783 .gt. 2sigma(I) |
| wR_factor_gt | 0.1826 | Structure | |
| shift/su_max | lt.0.001 | solution | SHELXS-97 |
| shift/su_mean | lt.0.001 | refinement | SHELXL-97 |
Get PDB coordinates
Get CIF text
Get SHELXL INS file
Back to the structure index

Date: Jun.1998